Genvinset® HLA C*06


The Noveos™ technology is based on the detection of specific IgE in blood, using paramagnetic spherical microparticles in an ELISA assay, to determine sensitisation to food, animals, plants, etc.
When an allergen, a substance that the body recognises as foreign, comes into contact with B cells, it triggers a response that involves the production of allergen-specific IgE by plasma cells, and it is this IgE that Noveos™ detects.
It is a technology:
How does it work?
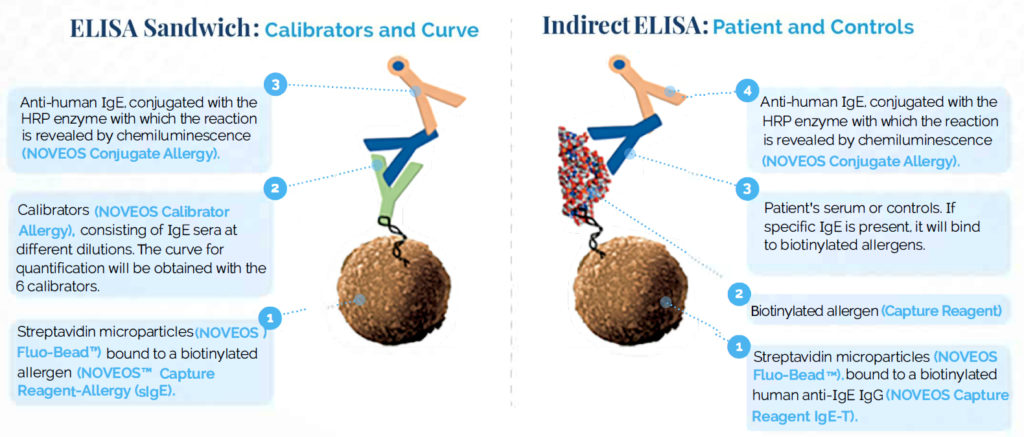
Normalisation is performed by quantifying the initial and final microparticles by fluorescence. The combination of fluorescence and chemiluminescence makes the test more accurate.
What does it allow us to do?
Assess specific IgE using whole allergen extracts (e.g. whole egg) or molecular components (e.g. egg white protein), even allowing the determination of the allergy trigger molecule, which is of interest to understand allergic cross-reactivity reactions.
Assess the clinical risk of allergic reaction. Thermostable proteins are indicative of a high risk of severe systemic allergic reaction, whereas thermolabile proteins produce mainly local reactions.
Identify the optimal immunotherapy treatments for each patient. Knowing the allergen that triggers the allergic reaction facilitates diagnosis and choice of appropriate treatment.
Features
Requires as little as 4 µL of sample per test, compared to 50-100 µL for other platforms.
Innovative and reliable technology
No false positives due to CCD (Cross-reactive Carbohydrate Determinants) and no false negatives due to interference with biotin in the sample (non-competitive indirect assay with paramagnetic microparticles).
Rapid and cost-effective technique
*Connected to water and waste.
For a list of available allergens, please contact us.
Public Documentation
Related products
2021 © Diagnóstica Longwood SL
This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Strictly Necessary Cookie should be enabled at all times so that we can save your preferences for cookie settings.
If you disable this cookie, we will not be able to save your preferences. This means that every time you visit this website you will need to enable or disable cookies again.
This website uses Google Analytics to collect anonymous information such as the number of visitors to the site, and the most popular pages.
Keeping this cookie enabled helps us to improve our website.
Please enable Strictly Necessary Cookies first so that we can save your preferences!
Más información sobre nuestra política de cookies
