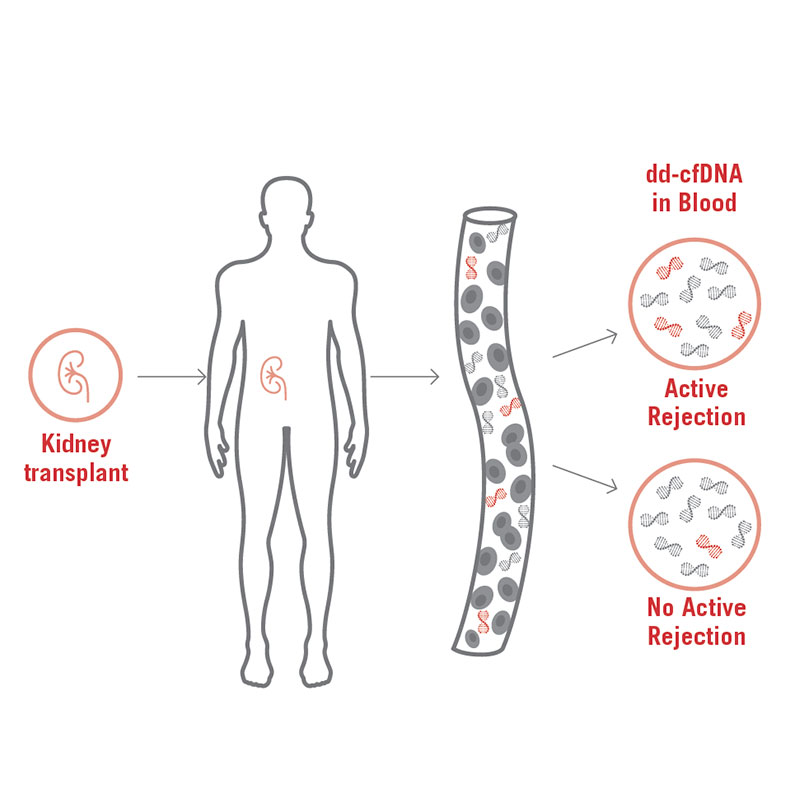
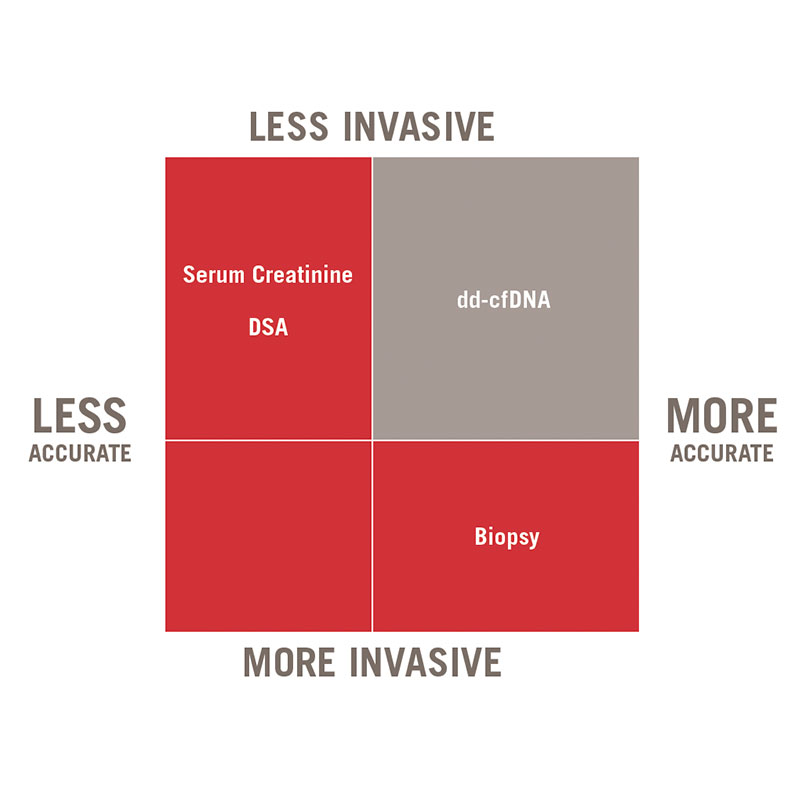
AlloSeq cfDNA is a kit that enables, in patients who have received a solid organ transplant, the relative quantitation of the cell free DNA (cfDNA) that originates from the graft using next generation sequencing. This quantitation allows the early detection of the graft injury and has demonstrated to be more sensible than serum creatinine in the early diagnose of renal rejection.
Furthermore, some studies have been published that show the utility of this test for monitoring the effectiveness of the treatment for acute rejection, as well as for the detection of the BK virus nephropathy.
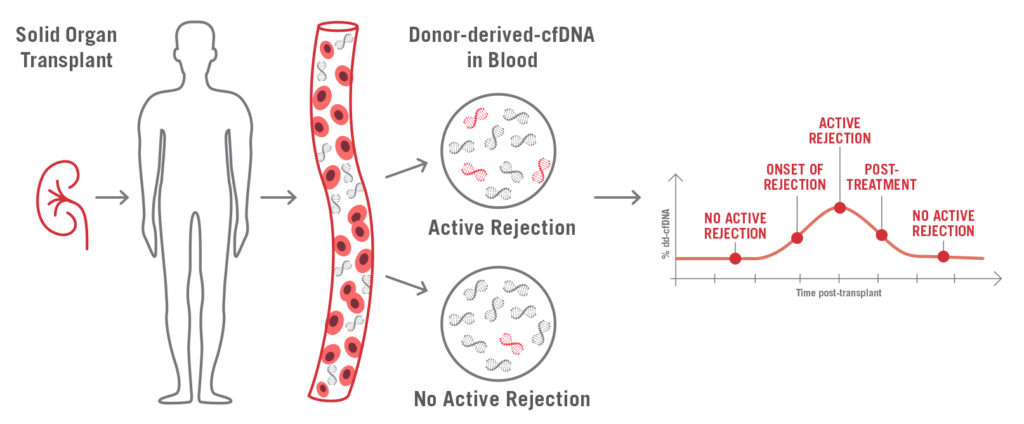
It’s a non-invasive test, since the recommended sample is plasma, and the results can be obtained in less than 24 hours. It uses 202 SNPs to precisely quantify the proportion of donor derived cfDNA.
The library preparation can be performed in less than 3 hours and it consists in a multiplex PCR in which the SNP-specific and index PCRs occur in the same reaction. Indexed samples are subsequently pooled together to prepare for purification and sequencing.
The data is analyzed automatically by a software, and the results are shown as the percentage of the donor-derived cfDNA.